How NBI Works
Olympus Narrow Band Imaging (NBI) is an optical technology available for a variety of medical disciplines which helps to visualize the minutest vascular and mucosal patterns. NBI uses only wavelengths absorbed by hemoglobin for maximum contrast. A number of studies highlight the clinical value of NBI, especially with regard to the detection of cancer and characterization of suspicious mucosal areas. Compared to white-light endoscopy, the images of capillaries are less blurred and the probability of missing a lesion is reduced.
The Technical Principle: Tissue Absorption
Normal white light consists of a specific number of wavelengths and is used as standard during cystoscopy. NBI light from Olympus works differently, revealing considerably more details about the tissue being examined.
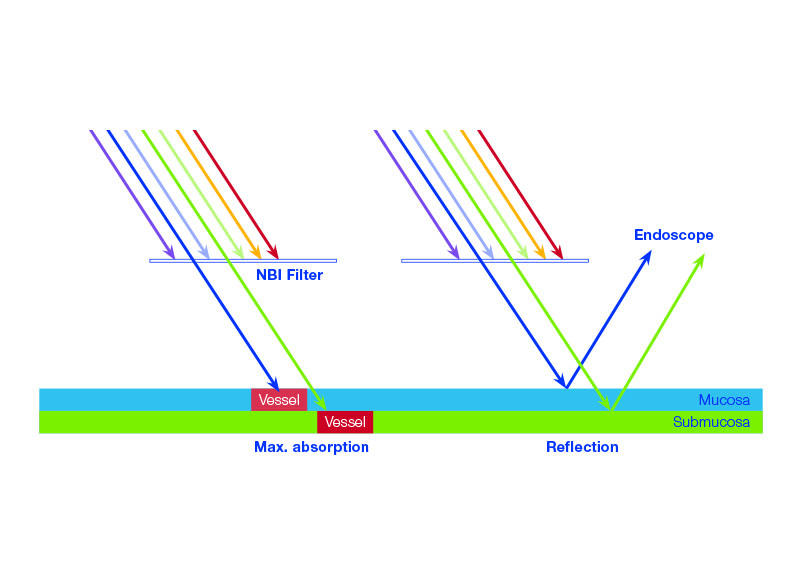
NBI light passes through a special narrow-band filter before it hits the tissue. The filter eliminates all other wavelengths, with the exception of the shorter blue and the longer green wavelengths.
When the blue and the green wavelengths hit the tissue, the light is absorbed by the hemoglobin contained in the blood vessels. The surrounding mucosal areas reflect the light back to the endoscope. As a result, the contrast of the blood vessels in relation to the mucosa is significantly enhanced. That makes the mucosal and vascular structure much clearer. In other words, because tumors are typically indicated by specific vessel structures, NBI light helps identify them and understand their pathology earlier on.
How does NBI help medical specialists?
Thanks to a large amount of clinical evidence, NBI really opens up many possibilities for diagnosis throughout a hospital. The high contrast between vessels and the surrounding mucosa means NBI facilitates the detection and characterization of tumors and even displays highly vascularized suspect lesions in deeper tissue layers. Compared with white-light endoscopy, the images of capillaries are less blurred and the probability of missing a lesion is reduced. As such, NBI improves specialists’ ability to reach an early and precise optical diagnosis.
NBI light exploits specific properties of light
The blue wavelength only penetrates the superficial layers of the mucosa and is absorbed by the vessels on the surface. The green wavelength is reflected, hence the green color of NBI images. That makes the tissue’s superficial structure much clearer and more visible to specialists, thus facilitating the detection of highly vascularized tumors.
Conversely, the green wavelength is absorbed by blood vessels within deeper mucosal layers. This is particularly helpful for displaying highly vascularized suspect lesions in deeper tissue layers. In addition, with NBI light the visualization of the capillary system is less blurred or distorted by scattering compared to white-light endoscopy.
In the end, this makes a difference: because NBI light is absorbed by vessels but reflected by mucosa, NBI light achieves the maximum contrast of vessels and mucosa. Therefore NBI sets a new standard in endoscopy and opens up many diagnostic possibilities that improve a specialist’s ability to reach an early and precise optical diagnosis of tumors.
Want to know the ins and outs of how NBI works?
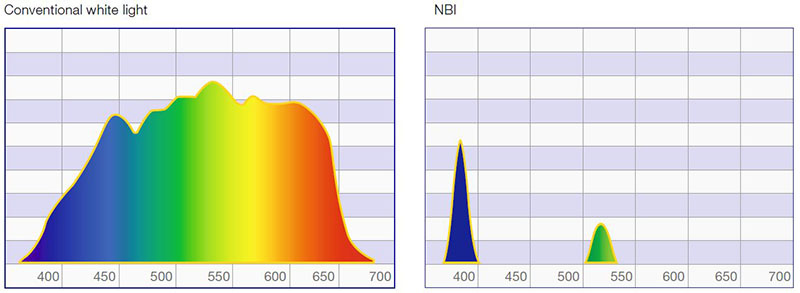
Normal white light consists of light of different wavelengths. But only a small part of the spectrum is visible – the part the human eye perceives as colors. When an object is illuminated by white light, some of the colors are absorbed while others are reflected. The reflected colors are the ones we see. White-light endoscopy has been proven to give an excellent view of vessel structures and the surrounding mucosa, especially with HDTV. But there is still room for improvement, and that comes with Olympus NBI light.
Olympus NBI light passes through a special narrow-band filter before it hits the tissue. The filter eliminates all wavelengths except the shorter 415 nm blue and the longer 540 nm green ones. Both blue and green wavelengths match exactly the absorption spectrum of hemoglobin. So when the blue and the green wavelengths hit the tissue, they are selectively absorbed by the hemoglobin and reflected by other structures.
Esophagus
This conclusion arises from an international, randomized, crossover trial comparing HD-WLE and NBI with 123 patients by Sharma et al. , published in Gut. 2013, 62 (1), pp. 15–21.
The aim of the study was to compare high-definition white-light endoscopy (HD-WLE) applying the Seattle protocol and NBI targeted biopsy for detection of IM and neoplastic tissue in Barrett’s Esophagus. The authors examined the differences of HD-WLE and NBI biopsies with respect to (1) the proportion of patients with intestinal metaplasia and neoplasia; (2) the proportion of neoplastic area; and (3) the number of overall biopsies performed.
The results: For detection of IM, HD-WLE and NBI each yielded detection rates of 92%. While HD-WLE required a mean of 7.6 biopsies per patient, NBI only required 3.6 biopsies. For detection of dysplasia, the diagnostic yield of HD-WLE and NBI were equivalent. However, NBI required fewer biopsies than HD-WLE in patients with short-segment BO (3.0 vs. 3.9) and patients with long-segment BO (4.1 vs. 10.9).
Thus, NBI may improve the efficiency of Barrett’s Esophagus endoscopic screening and surveillance and even reduce pathology costs, thanks to fewer biopsies being taken.
This result was found by Manabu Muto et al.in a randomized controlled clinical study with 360 patients, comparing the real-time detection rate of superficial SCC and HNSCC with WLE and NBI in a back-to-back fashion. See details in the Journal of Clinical Oncology. 2010, 28 (9), pp. 1566–1572.
Esophageal cancer is the eighth most common cancer in the world with mostly a poor prognosis. This is mainly due to the fact that white-light endoscopy has a poor detection rate of early-stage cancers. Therefore, esophageal SCC and head & neck SCC (HNSCC) are often detected at a late stage. The aim of the study was to verify if NBI could improve the detection rates for squamous cell carcinoma of the head and neck region as well as the esophagus.
The results were promising. While primary NBI detected all (100%) of the superficial cancers in the H&N region, primary WLE detected only 8.0% (Table 1). In the esophagus, primary NBI detected 97% of the lesions while primary WLE reached only 55%. The detection rate of secondary NBI after primary WLE significantly increased in both the H&N region (8.0% vs. 77%) and esophagus (55% vs. 95%). In contrast, if NBI was followed by secondary WLE, the detection rate declined. Fifty-seven percent of superficial cancers in the H&N region and 23% in the esophagus even were detected only by NBI. There was only one lesion that was detected by WLE but was missed by secondary NBI.
In conclusion, NBI has a significantly higher detection rate for SCC than white-light endoscopy and may become the standard examination for the early detection of superficial cancer in the H&N region and the esophagus.
Colon
A meta-analysis byMcGill et al.published in Gut 2013; 62 : 1704–1713 analyzed 28 studies on the real-time diagnostic performance of NBI colonoscopy. They found that real-time endoscopic diagnosis of colorectal polyps with NBI is highly accurate, with the area under the summary receiver operator curve exceeding >0.90. High-confidence predictions provide >90% sensitivity and NPV for adenomateous histology. Furthermore, surveillance interval predictions based upon the optical diagnosis were in agreement with pathology in more than 90% of patients.
In conclusion, the adaption of NBI optical diagnosis of colorectal polyps, particularly in high-confidence predictions, is promising for making colonoscopy more cost effective and efficient by potentially avoiding the need for pathological examination.
Hewett et al.Hewett et al. developed a simple tool to assess the histology of colorectal polyps: the NBI International Colorectal Endoscopic (NICE) Classification. Their randomized controlled multi-center study, published in Gastroenterology. 2012, 143 (3), pp. 599–607, does not only provide a simple tool for NBI-supported real-time assessment of colorectal polyps. It also shows that applying this classification scheme is accurate enough to consider the resect-and-discard policy which is a promising practice to save pathology costs.
The criteria of the NICE classification were rated both with experts and fellows followed by an overall prediction of histology. Surface pattern reached the highest values in accuracy, sensitivity, specificity, and negative predictive value. When combining the criteria, the presence of adenomateous features achieved an overall accuracy, sensitivity, and specificity of 92%, 92% and 95% respectively. The negative predictive value for adenomateous histology was 92%. These values further increased when low-confidence predictions were excluded.
In conclusion, if used with high-definition endoscopy, the NICE classification is suitable to differentiate between hyperplastic and adenomatous polyps.
Fulfilling the minimum performance benchmarks for assessing the histology of diminutive colorectal polyps, it may substantially contribute to reduce the cost of colonoscopy.
- Content Type