Author

Sang-Wun Kim, MD, PhD.
Professor, Department of Obstetrics and Gynecology, Director of Women’s Cancer Center of Yonsei Cancer Center, Yonsei University College of Medicine, Seoul, Korea.
Biography
Dr. Sang-Wun Kim graduated from Yonsei University College of Medicine in 1997, where he also earned his PhD degree in 2009. He completed his surgical residency training of Obstetrics and Gynecology in Severance Hospital in 2002. He was a researcher as military service in the division of genetic diseases of Korean National Institute of Health in 2002-2005, and a visiting scholar in Sanford-Burnham Medical Research Institute, San Diego, USA in 2012-2013.
He was the secretary general in the Korean Society of Obstetrics and Gynecology, in 2009-2011. He is currently a member of the American Association of Gynecologic Laparoscopists (AAGL), American Association of Cancer Research (AACR) and Korean Society of Gynecologic Oncology and Colposcopy (KSGOC). In 2014, he won the best surgical film award in the 24th Annual Meeting of Korean Society of Gynecologic Endoscopy and Minimally Invasive Surgery for the subject of Technical Tips for Single-port Laparoscopic Para-aortic Lymph Node Dissection using Conventional Laparoscopic Instruments.
Infrared observation in gynecology
Infrared (IR) observation using indocyanine green (ICG) is the most useful in the examination of the sentinel lymph node (SLN) for endometrial cancer among gynecologic cancers, although it is also used in the detection of cervical cancer and vulvar cancer. There are some case reports of its use in early ovarian cancer; however, its use has been largely experimental. The concept of SLN has been introduced in vulvar cancer since decades. In vulvar cancer, even before IR observation, a blue dye was first used as a standard, but it was not widely used as its applicability was difficult in practice.
However, IR observation has become a standard practice for the detection of the SLN for vulvar cancer. Since endometrial cancer is the most common cancer in western countries, ICG has been most commonly used for the detection of endometrial cancer. In Korea, the incidence of endometrial cancer used to be around a few patients a year, but recently, it seems to have increased to almost the same extent as that of cervical cancer.
Laparoscopic sentinel lymph node biopsy for endometrial cancer using IR observation
Surgical staging of endometrial cancers
When a patient with endometrial cancer is referred from the clinic, he or she first undergoes imaging tests such as magnetic resonance imaging (MRI) and positron emission tomography (PET)-computed tomography (CT) to determine the stage of the endometrial cancer.
Endometrial cancer is usually found in the body of the uterus and to a lesser extent, in the region of the cervix. If the endome1trium alone has cancerous lesions, endometrial cancer is stage 1. If the endometrial cancer has invaded the uterine lining, the uterine wall, and the uterine cervix, the endometrial cancer is the stage 2, and if it extents to the ovary and the ovarian ducts, the endometrial cancer is early stage 3; if it has spread up to the pelvis and lymph node, the endometrial cancer is late stage 3. Clinical staging is performed for differential diagnosis of the disease, whereas surgical staging is performed to evaluate the extent of metastasis. Most endometrial cancer cases are detected at an early stage, with 70%–80% of cases having lesions confined to the uterus. Patients whose cancer has metastasized are administered different treatments; 10%–20% patients show metastasis to nearby lymph nodes (pelvis or aortic lymph nodes), while very few patients show metastasis to other organs.
Metastasis takes place via several routes; hematogenous metastasis occurs along the blood vessels, while lymphogenous metastasis occurs along the lymphatic ducts. Thus, the characteristics of metastasis varies depending on the type of cancer. Endometrial cancer rarely spreads along the blood vessels and mostly spreads via the lymphatic fluids in the lymphatic ducts.
Therefore, even in early stages of endometrial cancer, one cannot confidently predict the risk of recurrence of the cancer after removing the uterus. This is because the cancer might have already spread to the lymphatic ducts and other lymph nodes. There is no way you can tell if it has metastasized without performing a lymph node biopsy. Predicting this risk of metastasis through imaging tests may lead to false-negative and falsepositive results. Hence, cancer can be diagnosed through imaging tests (clinical staging); however, detecting metastasis requires surgical staging.
If there is metastatic cancer, the endometrial cancer will not be stage 1 but stage 3, whose treatment is completely different as the patient would undergo radiotherapy and/or chemotherapy after surgery. In the past, the lymph nodes in the pelvis, aorta, and other parts of the body where there could be metastases were removed. Hence, we had to endure the risks of longer operation time and of major blood loss as well as the development of lymphedema and lymphoma. We had to dissect all the lymph nodes since we did not know where endometrial cancer spread from the uterus. I wish I knew where to go, but there was no way to know.
Sentinel lymph node biopsy using ICG
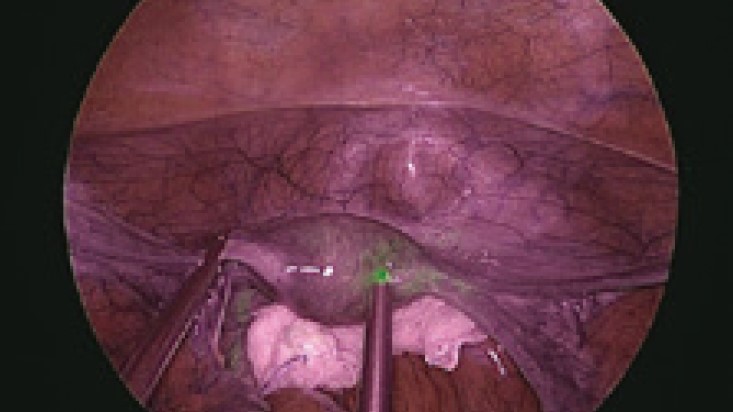
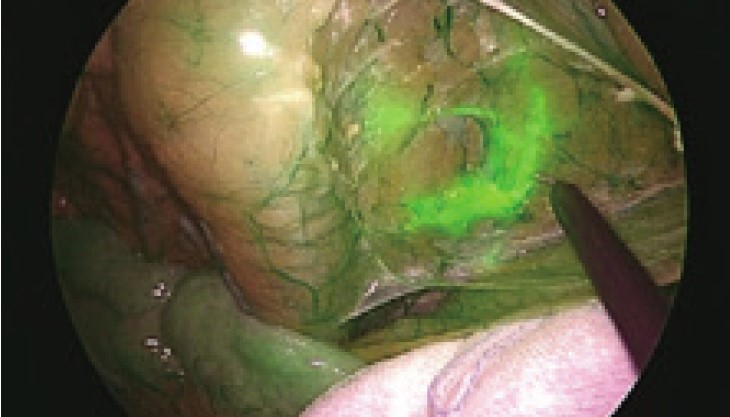
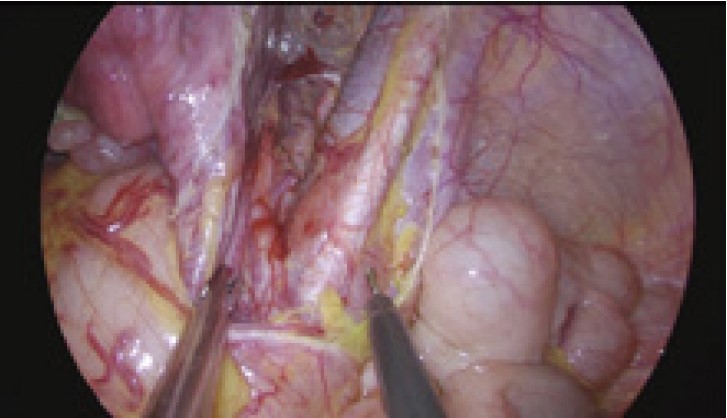
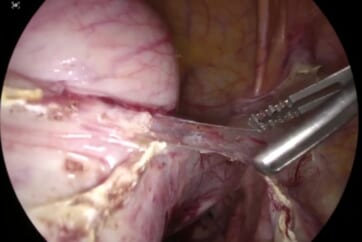
The reason we use ICG in IR observation is because if we inject ICG into the uterus, we can trace all the lymphatic ducts coming out of the uterus and see them clearly. After injecting ICG, following the lymphatic ducts leads us to lymph nodes that are like rest areas on the highway. When the first lymph node along the lymphatic duct is dissected from the uterus and examined, if there are no cancer cells in it, there is little chance of cancer in the following connected lymph node. Therefore, this first lymph node is called the SLN.
If you excise only the SLN, you can see immediately whether there is a metastasis to lymph nodes or not. You do not need to excise all the lymph nodes, but you need to selectively excise some of them off. The biggest advantage of IR observation using ICG is that it can reduce the risk of developing postoperative complications such as lymphedema or lymphocele. It also obviates other problems such as the risk of bleeding during surgery and long operating time.
Two-step SLN mapping strategy using ICG
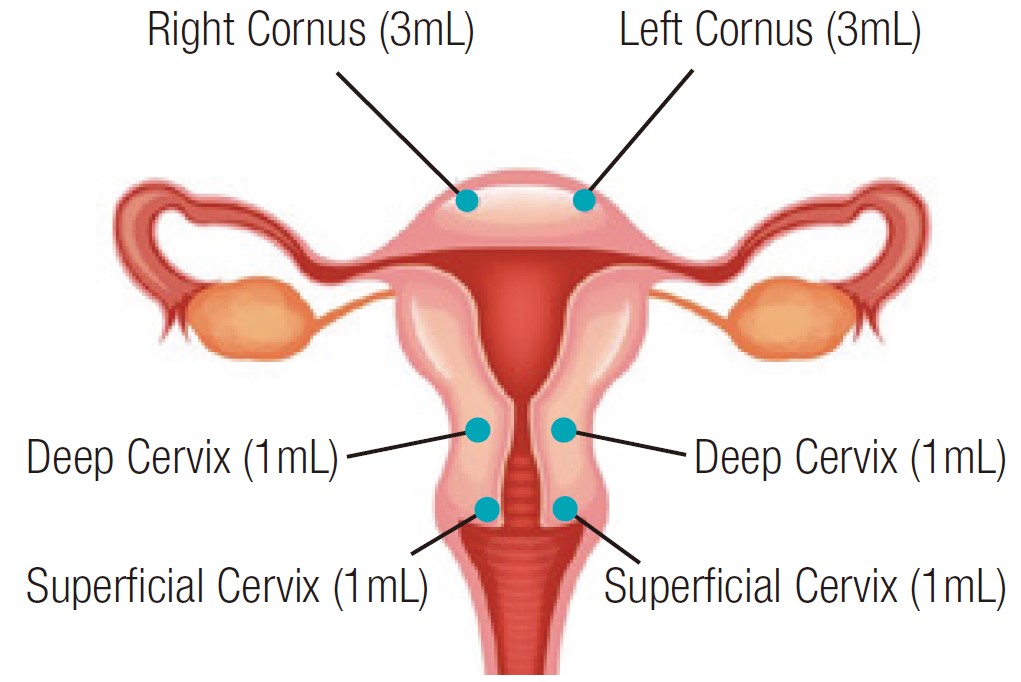
The current standard procedure of SLN mapping in endometrial cancer is the injection of ICG into the cervix. Injecting ICG makes it to spread mainly along the lymphatic ducts, and when the ICG spreads, all lymphatic ducts can be traced. The two-step SLN mapping strategy is to inject ICG into two parts of the uterus and see the lymph node channels spreading from the uterus. Three milliliter of ICG is injected into the cervix at 3 and 9 o’clock (Figure 1). The lymphatic fluid drains from the cervix towards the pelvis. For the upper part of the body of uterus, however, these lymphatic vessels, which travel with blood vessels, drain towards the perivascular lymph nodes around the aorta and the vena cavae.
Usually, it takes a lot of time to dissect all the lymph nodes and this increases the risk of severe bleeding. Surgically removing the SLN is not applied to every patient, but it has been used in the early stages of endometrial cancer, when lymph node metastasis is not clear. However, if there is obvious lymph node metastasis, the lymph node(s) should be excised. About 5 minutes after injecting ICG, you can clearly see the lymphatic ducts and they can remain visible for up to 1–2 hours.
Full lymph node dissection using ICG
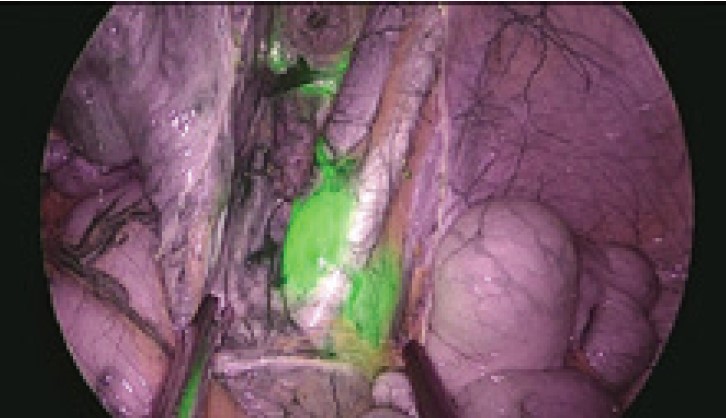
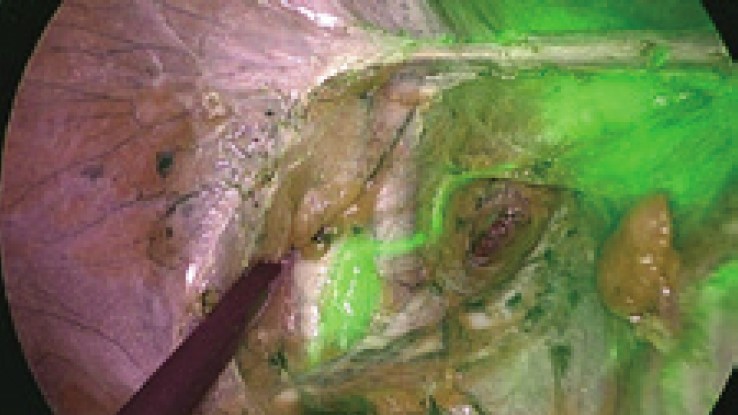
While most IR observations using ICG have been used for Sentinel lymph node detection until now, they may also be useful in determining the extent of lymph nodes dissection even if lymph node metastasis is clearly present and the lymph nodes need to be removed. After injecting ICG into the cervix, all the lymphatic ducts that come out of the uterus can be traceable in a little time. When all the lymph nodes around the uterus become visible, they can be excised, under the guidance of fluorescent-image, especially when there is a possibility of lymph node metastasis requiring.
Before injecting ICG, you do not know where the fat tissue is, where the lymphatic duct is, or where the lymph nodes are. If you wait for some time after injecting ICG, you will be able to see the lymph nodes and the lymph ducts. Therefore, you can perform surgery while actually checking your patient’s surgical staging and seeing how far the surgery would go. Laparoscopic surgery to remove the lymph nodes around the aorta and entire lymph nodes in the pelvis is more difficult than open abdominal surgery. Therefore, image-guided complete lymphadenectomy can also use IR observation using ICG.
Tips and precautions while using IR observation for SLN biopsy
When injecting ICG, the injection site may not have been properly selected. The ICG can also be wrongly injected. In both scenarios, it is permitted to repeat the injection procedure and observe the outcome. Inexperienced surgeons usually inject it into the cervix and see the SLN in the pelvis, and if you open the lymph nodes around it in advance, the lymph nodes in the pelvis are all broken. Because the lymphatic ducts are usually thin and may burst even to the
slightest touch, ICG may spill and contaminate surrounding tissues, making it difficult to distinguish the lymph nodes. To avoid it, you need to inject an ICG and then, after some time, carefully open up the peritoneum to find the lymph nodes and the lymph ducts.
The branching pattern of lymphatic vessels is complex, as lymphatic ducts may bypass the adjacent lymph node and drain into another lymph node.
However, the first lymph node usually becomes green before the next lymph node. Thus, many lymph nodes are stained, only the first one is the true SLN.
Hence, even if they are fluorescent through ICG staining, they are not all SLNs. Only the SLN should be excised.
References
1. Eoh KJ, Lee YJ, Kim HS, Lee JY, Nam EJ, Kim S, Kim YT, Kim SW. Two-step sentinel lymph node mapping strategy in endometrial cancer staging using fluorescent imaging: A novel sentinel lymph node tracer injection procedure. Surg Oncol. 2018 Sep;27(3):514-519.
2. Nam EJ, Kim SW, Kim S, et al. A Case-Control Study of Robotic Radical Hysterectomy and Pelvic Lymphadenectomy Using 3 Robotic Arms Compared With Abdominal Radical Hysterectomy in Cervical Cancer. International Journal of Gynecologic Cancer 2010;20:1284-1289.
- Content Type