Laparoscopic Anatomical Segment 8 Segmentectomy

Prof. Brice Gayet, MD, PHD
Professor of Digestive Surgery at Paris Descartes University Medical School
Head of the Digestive Surgery Department of the Montsouris Institute in Paris, France.
Situation in Europe
The spreading of laparoscopic liver resections (LLR) is still limited in Europe as well as in France despite the presence of a few world-renowned expert centers. Overall, only 20% of hepatectomies are performed by laparoscopy, and over 50% are left lateral sectionectomies. A paper focused on major hepatectomies in 10 European centers reported a rate of 22% of major resections on 2245 LLR. A recent meta-analysis on Hepatocellular carcinoma (HCC) in Europe was able to find around 600 cases done by laparoscopy. In conclusion, experienced teams in Europe have shown the possibility of performing all types of resection with a minimally invasive approach provided that the patients are selected according to the skills of the surgeon.
Advantages and Risk of Laparoscopic Approach
Laparoscopy has many advantages, some of which are not dependent on the procedure or the pathology, such as the reduction of postoperative pain, bleeding and length of hospital stay. This is also true for hepatectomies. Laparoscopy reduced bleeding and the need for transfusions in all randomized or cohort studies. This is perhaps due to an improved view of the surgical site, particularly at the upper and posterior portion of the liver, or due to the counterpressure apply by the pneumoperitoneum at the dissection.
However, liver surgery can be life-threatening. The first risk factor for postoperative death is liver failure due to the quality of the remaining liver, and this risk is not affected by the laparoscopic approach. This complication is most often due to the volume of the remaining liver being too small, and this therefore concerns major hepatectomies (more than 3 segments of the liver). The second is the risk of intraoperative death by bleeding or air embolism, and hepatectomy is virtually the only intervention in gastrointestinal surgery which presents this risk. This risk is related more to the complexity of the procedure than to the volume of the resection, and this therefore concerns complex hepatectomies. We classified laparoscopic procedures according to their complexity (Ref 1 Kawaguchi). It appears essential at the beginning of the experience to proceed with the classification step by step, and to only advance to the higher stage after mastering the preceding one. The report presented here is about segment 8 segmentectomy – a minor but complex procedure.
Theoretical challenges related to laparoscopy
Loss of 3D vision for surgery in a large-volume organ, the largest organ of the body, theoretically increases the risk of missing the correct sectional plane or misjudging the anatomical limits of a liver segment. This loss was only offset by the expertise of the surgeon. Currently, 3D cameras with selective dye portal injections or even the use of Indocyanine green (ICG) with infrared cameras can compensate for this limitation. Flexible optics (ENDOEYE Flex 3D) also provide the advantage of being able to view all the sides of the liver more easily.
Access to the postero-superior liver segments (S1, S4a, S7 and S8). The liver is an organ situated entirely under the ribs. The fact that the cameras make it possible to view the patient’s body from the inside, under the ribs in any case, allows for a better overall visualization. For the postero-superior segments, it is possible to use flexible optics as we did or to place the camera and an operating trocar across the diaphragm to approach the liver from above, as proposed by Prof. Makuuchi who essentially suggests using thoracotomy for difficult cases on the right side. In laparoscopy it is sometimes also necessary to use the tilt of the operating table to expose the posterior right side of the liver.
The lack of haptic feedback is currently compensated only by the surgeon’s experience. To identify tumors, ultrasound and sometimes echo-elastography as well as infrared cameras are usually sufficient. However, robotic tools are being evaluated and should also, in a near future, compensate for the lack by providing greater accuracy of the procedures thanks to haptic feedback.
Air embolisms are a reality. Gas bubbles entering the veins is a real danger when cutting the hepatic parenchyma whether it is done by open surgery or a minimally invasive procedure. This risk has been considered by some as a reason not to use laparoscopy because of the additional risk due to the pneumoperitoneum. Effectively animal studies demonstrate that CO2 embolism was found in 19 of the 26 cases of laparoscopic liver resection.
The greatest danger during laparotomy is actually air embolisms, which are dangerous because the air consists of oxygen (20%), which is poorly soluble in blood (solubility: 0.02) and nitrogen (80%), which is even less soluble (0.0005). In contrast, the pneumoperitoneum is composed of CO2, an extremely soluble gas (0.5), and the risk of symptomatic embolism is therefore particularly low. According to a literature review, 0.2% of gas embolisms were reported among patients who underwent laparoscopic major hepatectomy without mortality reported. This is also why we ask anesthesiologists to report any increase in pCO2 that would signify the opening of a hepatic vein or of the inferior vena cava. Unlike venous wounds made during a laparotomy, which bleed profusely, in laparoscopy these wounds might not bleed due to the intra-abdominal pressure which is greater than the central venous pressure. This is also why we never use argon beam coagulation in laparoscopy. Argon is a gas which is as insoluble as nitrogen, with several deaths reported in laparoscopy possibly due to intraabdominal hyperpressure favoring embolism, with the standard mode of argon insufflator reaching 14 liters/minute.
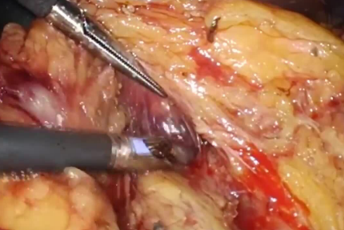
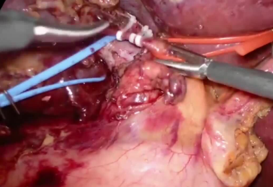
Bleeding is every surgeon’s fear during a hepatectomy. During the dissection of the hepatic pedicle, there is rarely an arterial or portal wound secondary to anatomical variations in the hepatic pedicle that are not recognized by the surgeon during preoperative examinations. Overall, they are easy to monitor. Massive hemorrhages can sometimes occur when cutting through the hepatic parenchyma because the liver is a hypervascular organ with three entangled branches of the arterial tree – the intrahepatic and portal veins on one side and the suprahepatic vein on the other side. Wedge resections pass through its networks and cut vessels in a practically unpredictable manner, while anatomical excisions based on hepatic segmentation occur along the hepatic veins that are precisely located between the segments.
The principles for reducing this bleeding are identical during both open surgery and minimally invasive methods. The aim is to keep the central venous pressure (CVP) low on the anesthetist side by a minimal filling and to lower or even suppress the blood supply into the liver by clamping the hepatic pedicle either en masse (Pringle) or electively at the level of the glissonian pedicles. In the case of acute bleeding, one should visualize the vascular breach for selective examination, alternating suction, irrigation and compression.
There are two major liver dissection techniques, and in reality surgeons use whichever one is required:
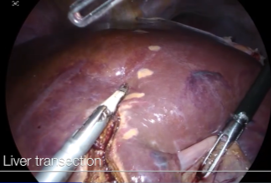
(a) The Kelly clamp crushing technique which involves crushing the parenchyma while respecting the vessels which can then be selectively controlled. It is traditionally performed using a Kelly or a Maryland, but it can be done with tools adding simultaneous hemostasis such as bipolar forceps, Ligasure™, Harmonic™ or THUNDERBEAT. The advantage of this last is that it can also cut or not according to the decision of the surgeon avoiding, in laparoscopy in particular the entrances and exits of several tools always risking to hurt on their way. The advantage of the latter is that it can cut or not as decided by the surgeon in order to avoid injury during the entry and exit of the tool, in particular in laparoscopy, which is always a risk with this type of instrument.
(b) Visualization by dissection of the vessels before their control.This is the purpose of Handy-jet or Waterjet dissectors or the much more commonly used Dissectron, Surgitron or Cusa ultrasonic dissectors. In theory, they should reduce bleeding, but this is not supported by the controlled studies. In laparoscopy, they often cause contamination of the optics and due to the necessary suction, they also cause variations in pressure that are detrimental to maintaining a stable intravascular pressure. We learned to use the THUNDERBEAT without pressing too much on the vascular walls by using the jaws of the THUNDERBEAT ajar to retract the small orifices of the numerous vessels that drain into the hepatic veins along their path. Combining THUNDERBEAT with a bipolar grasper therefore allows us to switch from one technique to another without changing the instruments. We no longer use ultrasonic surgical aspirator, which was our main tool at the beginning of our experience.
1) Positioning the patient, positioning the trocars
The patient is placed on their back with their legs apart and a little bent. The hemithorax is raised with cushions placed under the right shoulder and the left arm held in a splint above the head. Counter-supports are placed on either side of the torso, especially at pelvis level, in order to allow the table to be tilted to the right for the initial dissection, when the surgeon stands between the legs and the assistant on the right or left. Then the table is tilted to the left when the surgeon comes to the right side with the assistant placed between the legs.
Five trocars are used (two 12 mm and three 5 mm) with the option to add another 5 mm trocar in case of exposure difficulty. To begin the intervention, three of these are positioned in the abdomen parallel with the costal margin at a height dependent on the hepatic volume, and if required, two are then positioned in the intercostal area – one 12 mm usually in the 7th space to be decided after mobilization of the liver. These two trocars are inserted under the control of the endoscopic view by pushing the diaphragm upwards to facilitate introduction. We use balloon foam trocars at this level to avoid pneumothorax. If this occurs nevertheless, a small angiocatheter placed in the pleura is left open during the entire operation to prevent ballooning of the diaphragm. The 12 mm trocar is used for ultrasound scanning and for the possible positioning of large 10 mm or 12 mm clips at the beginning of the procedure and for the laparoscope during the second stage of the resection.
- Content Type